
Fractional Leadership To
Recapture Growth
The battle-tested marketing and sales executives CEOs need.

Your Fractional EdgeTM
CEOs need help growing their companies but don't always have the time or money to hire a full-time CMO or CSO. Recruiting a full-time executive can take months, not to mention the ongoing costs.
By partnering with Chief Outsiders, CEOs can quickly employ battle-tested CMOs and CSOs to build and execute their growth engines.
In today's fast-paced digital age, Artificial Intelligence (AI) is no longer a futuristic concept reserved for tech giants—it's a present-day game-changer for small and medium-sized businesses (SMBs). Find out how AI is transforming how SMBs grow.
Grow with the Industry Leader

The Growth Gears™
Chief Outsiders uses The Growth Gears™ framework. The three gears driving growth for marketing and sales are Insight, Strategy, and Execution.
Ready to grow in 2024? Request a Growth Gears Assessment to clarify your company's growth challenges and opportunities.
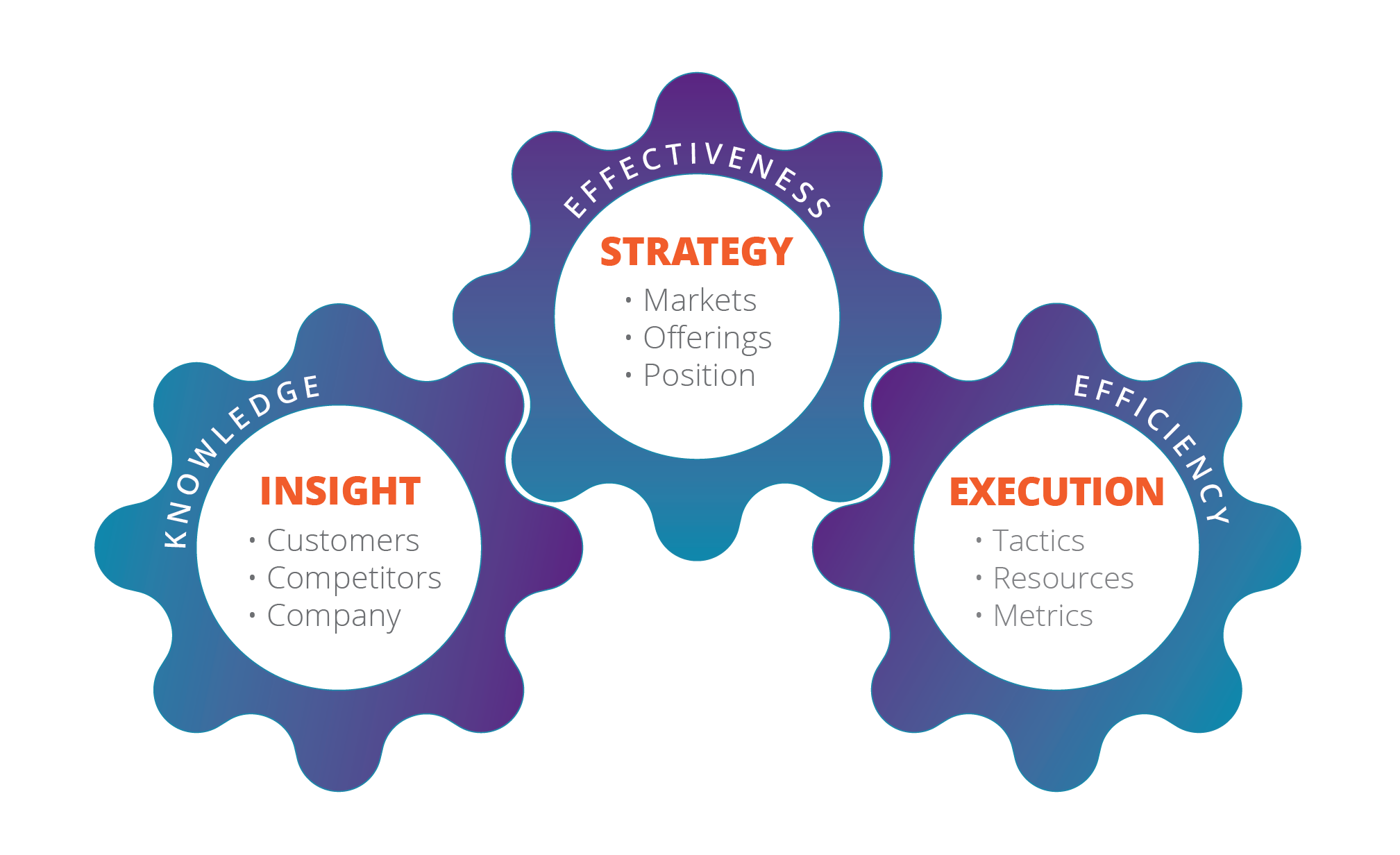

Insight
It’s important to continually seek out and make use of insights about your company, your customers, the marketplace, and your competition. These insights add up to a knowledge base from which you can make the right decisions to drive growth.

Strategy
Strategy is the second gear in the Growth Gears framework, and you create and maintain it using the knowledge gained from the first gear – Insight. Think of this not only as your marketing strategy but an overall business strategy – around your market, offerings, and positioning.

Execution
Once you have the right strategy, the third gear in the Growth Gears framework - Execution, is about implementing that strategy with the greatest level of efficiency. This involves efficient resource allocation, goal alignment, proactive decision-making, and operational excellence, driving measurable results and sustainable business growth.
What Our Clients Say:

A.J. Smith, Trystar, CEO
Partnering with Chief Outsiders was absolutely the right decision for Trystar! We achieved so much more than I expected in a relatively short period of time.

Dominick DeRosa, PELITAS, Chief Client Officer
We just had a board meeting where we reviewed the Chief Outsiders' engagement, and they couldn't have been happier. We achieved everything that we set out to achieve and our PE sponsors were thrilled.

Frank Ewing, AML RightSource, CEO
We've grown this company in the last two years from 450 to 1,000 FTEs. We couldn't have achieved that without the assistance from Chief Outsiders.
Featured Services
Positioning
Demand Generation
Sales Process
Digital Growth
Ecommerce
Team Development
Partner with Chief Outsiders
When you schedule a consultation with us, one of our Managing Partners will find the right fractional CMO or CSO for you.
How it works
Engaging with Chief Outsiders involves four steps, and each provides value.

Initial Consult

Fractional Match

Growth Plan

Quick Start

30 Day Money Back Guarantee
While we are confident that we will add value quickly and that you will be happy with your decision to engage Chief Outsiders, we recognize this is a significant investment and not without risk. Considering today’s uncertain and quickly changing business environment we offer you this Money Back Guarantee:
If at 30 days into the engagement you are not satisfied with the trajectory of the work that we are doing with your company, we will refund your engagement fees and stop any future billing. Travel or other out of pocket expenses including outside agency fees are not refundable.
From the Blog
Business Growth Insights for CEOs and Private Equity Executives
- Press Releases
- Careers
- Case Studies
- Marketing Consultant Company
- Marketing Strategy Consultants
- Marketing Plan Consultants
- B2B Marketing Consultants
- Virtual CMO
- Marketing Consultant Outsourcing
- Fractional CMO
- What is a Fractional CMO
- Healthcare Marketing Consultant
- Marketing Consultant Houston TX Texas
- Marketing Consultant Texas TX
- CEO Blog
- Ebooks Plus
- Webinars
- Executive Marketing Consultants
- Product Marketing Consultants
- B2C Marketing Consultants
- Virtual Marketing Consultants
- Senior Marketing Consultants
- Temporary CMO
- Hire a CMO
- Fractional CMO Salary
- Fractional CMO Responsibilities
- Marketing Consultant Austin TX Texas
- Marketing Consultant Dallas TX Texas
Houston, TX 77056
© 2023 Chief Outsiders






